Discussion
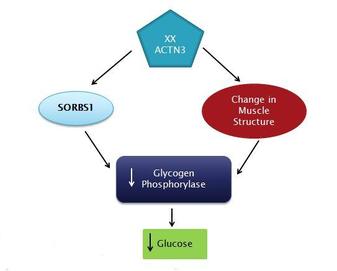
In addition to the speed phenotype, researchers now believe that ACTN3 may have influenced muscle metabolism. This theory was developed after researchers noticed there has been a strong recent selection for the non-functional ACTN3 X allele in certain populations (1). By studying a mouse model created by knocking out ACTN3 it was discovered that muscles lacking alph-actinin-3 uses energy more efficiently. Particularly, the decrease in glucose metabolism and an increase in beta-oxidation (2). This was coupled with the observation that patients with McArdle’s Disease (a disease in which the enzyme glycogen phosphorylase which breaks down glycogen to glucose is inactive) and the ACTN3 XX phenotype had improved exercise capacity (3). I propose to better study the role ACTN3 plays in the change in metabolic fuel found in these muscle fibers.
First Question
Question: Does climate,and therefore climate-related food availability, affect the ACTN3 protein across different species?
Hypothesis: That species in similar climates would have more similar ACTN3.
Hypothesis: That species in similar climates would have more similar ACTN3.
Experiment Procedure:
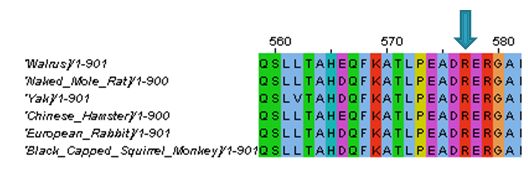
I tried to compare the ACTN3 protein sequence across several species in habitats ranging from polar and arid to temperate and tropical. I expected to see a change at amino acid 578 from Arginine to a stop codon in some of the animals found in more extreme climates (Figure 1). I did not however see this, after a literature search I discovered the the R to X mutation probably arose in the human population around 30,000 years ago (2). I also compared the domains and discovered that ACTN3 was very well conserved all the way down to the fruit fly and nematode (see protein domains). After looking at the phylogeny of animals from different climates, I concluded that the differences in ACTN3 across species were probably the result of something other than climate.
I tried to compare the ACTN3 protein sequence across several species in habitats ranging from polar and arid to temperate and tropical. I expected to see a change at amino acid 578 from Arginine to a stop codon in some of the animals found in more extreme climates (Figure 1). I did not however see this, after a literature search I discovered the the R to X mutation probably arose in the human population around 30,000 years ago (2). I also compared the domains and discovered that ACTN3 was very well conserved all the way down to the fruit fly and nematode (see protein domains). After looking at the phylogeny of animals from different climates, I concluded that the differences in ACTN3 across species were probably the result of something other than climate.
Second Question
Experimental Procedures:
1. Run a Microarray to check that the expression of glycogen phosphorylase (GPh) doesn't change in the XX phenotype. In the XX phenotype I would expect the expression of GPh to change because I think it is regulated post-translationally by ACTN3. I would expect there to be an increase in ACTN2 to correct for the non-functional ACTN3 and I would expect an increase in expression of enzymes involved with beta-oxidations such as Medium-chain acyl-CoA dehydrogenase.
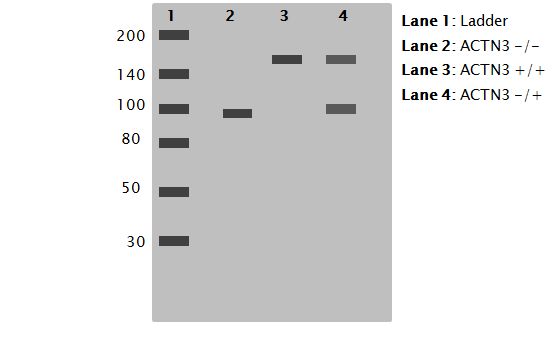
2. Using a Western Blot ensure that there is a change in GPh phosphorylation between XX and RR and RX genotypes (Figure 3). I would expect there to be decreased phosphorylation in the XX phenotype because of the change in muscle structure resulting in the change in phosphorylation site availability.
1. Run a Microarray to check that the expression of glycogen phosphorylase (GPh) doesn't change in the XX phenotype. In the XX phenotype I would expect the expression of GPh to change because I think it is regulated post-translationally by ACTN3. I would expect there to be an increase in ACTN2 to correct for the non-functional ACTN3 and I would expect an increase in expression of enzymes involved with beta-oxidations such as Medium-chain acyl-CoA dehydrogenase.
2. Using a Western Blot ensure that there is a change in GPh phosphorylation between XX and RR and RX genotypes (Figure 3). I would expect there to be decreased phosphorylation in the XX phenotype because of the change in muscle structure resulting in the change in phosphorylation site availability.
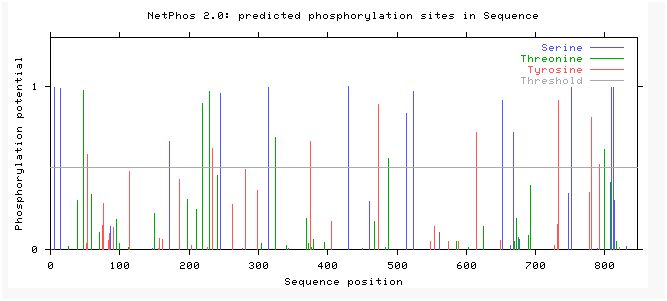
3. I then used NetPhos 2.0 to find the expected phosphorylation sites on the GPh protein (Figure 4). I was looking specifically for sites that tyrosine was phosphorylated because SORBS1 is involved with tyrosine phosphorylation. I thought that null ACTN3 may decrease SORBS1 activity therefore decreasing phosphorylation at these sites.
4. I would then mutate the Tyrosine Phosphorylation sites in a normal ACTN3 animal to decrease the phosphorylation of the GPh and compare the enzyme activity levels to the ACTN3 XX enzyme activity levels to try to determine where SORBS1 may be interacting with GPh.
Conclusions
We are currently faced with the daunting task of understanding how to combat all sort of modern diseases such as diabetes, cancer, obesity, and CVD. By looking back at how certain genes functioned in the past and how those genes helped our ancestors survived we can better understand how the disregulation of these genes play a role in many modern diseases.
By studying genes like ACTN3 we will better understand selective forces in our past and how, as humans, we have over come these forces. In a sense, we will gain better of understanding of what it means to be "human" and unique from our ancestors.
Future Directions
I think that there are several areas that would be interesting for future research involving ACTN3.
1. Study the relationship between ACTN3 XX and ethnic groups with history of famine,
2. The migration of ACTN3 X allele in relation to human migration out of Africa.
3. ACTN3 genotypes in relation to modern diseases
4. The effect of ACNT3 genotype on response to exercise and ageing.
5. The effect of ACTN3 and the onset and severity of muscle disease pheotype.
1. Study the relationship between ACTN3 XX and ethnic groups with history of famine,
2. The migration of ACTN3 X allele in relation to human migration out of Africa.
3. ACTN3 genotypes in relation to modern diseases
4. The effect of ACNT3 genotype on response to exercise and ageing.
5. The effect of ACTN3 and the onset and severity of muscle disease pheotype.
Finally, below you will find a copy of my final presentation in this class. Please feel free to contact with me any further questions or comments.
| Final Presentation | |
| File Size: | 2766 kb |
| File Type: | |
References
1. North KN, Yang N, Wattanasirichaigoon D, Mills M, Easteal S, et al. (1999) A common nonsense mutation results in a-actinin-3 deficiency in the general population. Nature Genetics 21: 353–354.
2. North KN. (2008) Why is alpha-actinin-3 deficiency so common in the general population? The evolution of athletic performance. Twin Res Hum Genet. 2008 Aug;11(4):384-94. doi: 10.1375/twin.11.4.384.
3. Lucia, A., Gomez-Gallego, F., Santiago, C., Perez, M., Mate-Munoz, J. L., & Chamorro-Vina, C. (2007). The 577X allele of the ACTN3 gene is associated with improved exercise capacity in women with McArdle’s disease. Neuromuscular Disorders, 17, 603–610.
2. North KN. (2008) Why is alpha-actinin-3 deficiency so common in the general population? The evolution of athletic performance. Twin Res Hum Genet. 2008 Aug;11(4):384-94. doi: 10.1375/twin.11.4.384.
3. Lucia, A., Gomez-Gallego, F., Santiago, C., Perez, M., Mate-Munoz, J. L., & Chamorro-Vina, C. (2007). The 577X allele of the ACTN3 gene is associated with improved exercise capacity in women with McArdle’s disease. Neuromuscular Disorders, 17, 603–610.